To adhere or not to adhere? This is the disulfide exchange question!

Highlights by Marcela Franco Mineiro, PhD, from Instituto de Química da USP
Marcela got her PhD in 2019 under supervision of Flavia Carla Meotti at the Laboratory of Redox Processes in Inflammation, Department of Biochemistry, Institute of Chemistry, University of São Paulo, Brazil
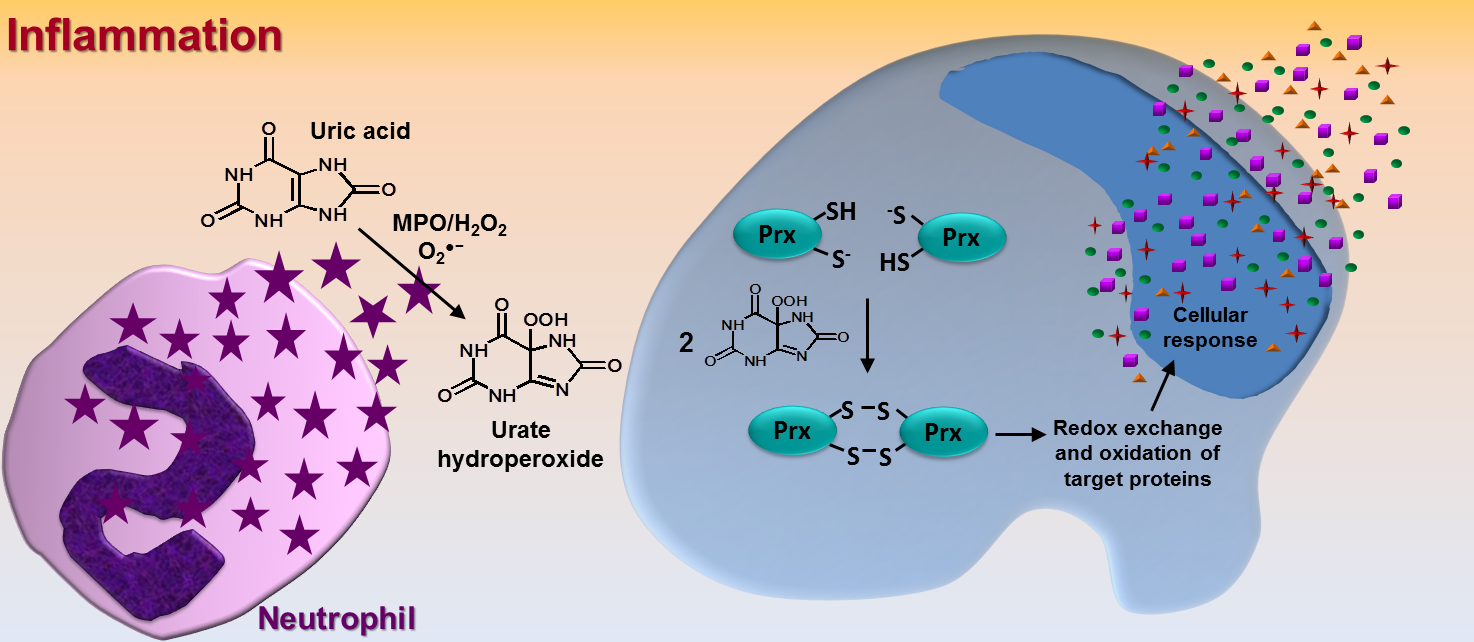
The term ‘adhesion’ is simply defined as ‘steady or firm attachment’ [1]. In cell biology, however, the adhesion process is far more complex and involves countless well-orchestrated reactions and interactions between proteins at both extra and intracellular surface of the plasma membrane. Intra and intermolecular disulfide exchange in these proteins is one of the most relevant mechanism that drives cell adhesion/detachment. Therefore, oxidizing and reducing agents can directly affect cell adhesion. Nonetheless, it is hard to infer whether an oxidant would increase or decrease this phenomenon because adhesion involves disulfide linkage/break at specific cysteine residues within each protein. It is well known that catalytic cysteines from extracellular cell surface protein disulfide isomerases (PDIs, of which the prototype is PDIA1, refered to as PDI) are crucial in cell adhesion and disulfide exchange between PDI and integrin takes place during the adhesion process [2, 3]. Of relevance, our group has shown that cysteines at the catalytic site of PDI are rapidly oxidized by urate hydroperoxide (6 × 10³ M⁻¹s⁻¹) [4]; this is a peroxide generated from uric acid in the inflammatory oxidative burst [5]. In fact, the oxidation of PDI by urate hydroperoxide is much faster than that by glutathione disulfide (188 M⁻¹s⁻¹) [6] or hydrogen peroxide (17.3 M⁻¹s⁻¹) [7], but slower than the oxidation by peroxynitrite (6.9 × 10⁴ M⁻¹s⁻¹) [7]. We found that the oxidation of extracellular cell surface PDI by urate hydroperoxide impaired the adherence of vascular endothelial cells to fibronectin in the same way as the thiol alkylating p-CMBS, the PDI inhibitor Rutin and the integrin blocking peptide RGDS. Interestingly, adhesion was markedly inhibited in the first 30 min and, to the exception of the treatment with the irreversibly thiol alkylating p-CMBS, cells were able to adhere after 90 min of treatment [4]. These results show that the oxidation of thiols and inhibition of PDI or integrin disrupt cellular adhesion in a transient way. However, the continuous production of oxidants, as in vascular inflammation, might further recover cell adhesion. Since urate hydroperoxide can be formed extracellularly and efficiently targets cell surface PDI affecting cell adhesion, it might be a mechanism underlying the known vascular endothelial dysfunction described for uric acid. Analogous effects of other vascular oxidants in cell adhesion remain to be investigated.
References
- Merriam-Webster [on-line] 2020. | url: https://www.merriam-webster.com/dictionary/adhesion
- N. Rosenberg, R. Mor-Cohen, V. H. Sheptovitsky, O. Romanenco, O. Hess, J. Lahav. Integrin-mediated cell adhesion requires extracellular disulfide exchange regulated by protein disulfide isomerase Experimental Cell Research, 381(1): 77–85, 2019. | doi: 10.1016/j.yexcr.2019.04.017
- A. I. Soares Moretti, F. R. Martins Laurindo. Protein disulfide isomerases: Redox connections in and out of the endoplasmic reticulum Archives of Biochemistry and Biophysics, 617: 106–19, 2017. | doi: 10.1016/j.abb.2016.11.007
- M. F. Mineiro, E. de S. Patricio, Á. S. Peixoto, T. L. S. Araujo, R. P. da Silva, A. I. S. Moretti, F. S. Lima, F. R. M. Laurindo, F. C. Meotti. Urate hydroperoxide oxidizes endothelial cell surface protein disulfide isomerase-A1 and impairs adherence Biochimica et Biophysica Acta (BBA) - General Subjects, 1864(3): 129481, 2020. | doi: 10.1016/j.bbagen.2019.129481
- R. P. Silva, L. A. Carvalho, E. S. Patricio, J. P. Bonifacio, A. B. Chaves-Filho, S. Miyamoto, F. C. Meotti. Identification of urate hydroperoxide in neutrophils: A novel pro-oxidant generated in inflammatory conditions Free Radical Biology and Medicine, 126: 177–86, 2018. | doi: 10.1016/j.freeradbiomed.2018.08.011
- Á. S. Peixoto, R. R. Geyer, A. Iqbal, D. R. Truzzi, A. I. Soares Moretti, F. R. M. Laurindo, O. Augusto. Peroxynitrite preferentially oxidizes the dithiol redox motifs of protein-disulfide isomerase Journal of Biological Chemistry, 293(4): 1450–65, 2017. | doi: 10.1074/jbc.m117.807016
- A. Lappi, L. W. Ruddock. Reexamination of the Role of Interplay between Glutathione and Protein Disulfide Isomerase Journal of Molecular Biology, 409(2): 238–49, 2011. | doi: 10.1016/j.jmb.2011.03.024
Add new comment